Describe How to Use Steel Wool to Remove Copper Ions
Steel wool on the other hand is made of lots of thin strands and so a lot more iron atoms are in contact with the oxygen in the air. When the copper is dry carefully dry the outside of the beaker and reweigh to find the mass of copper formed.
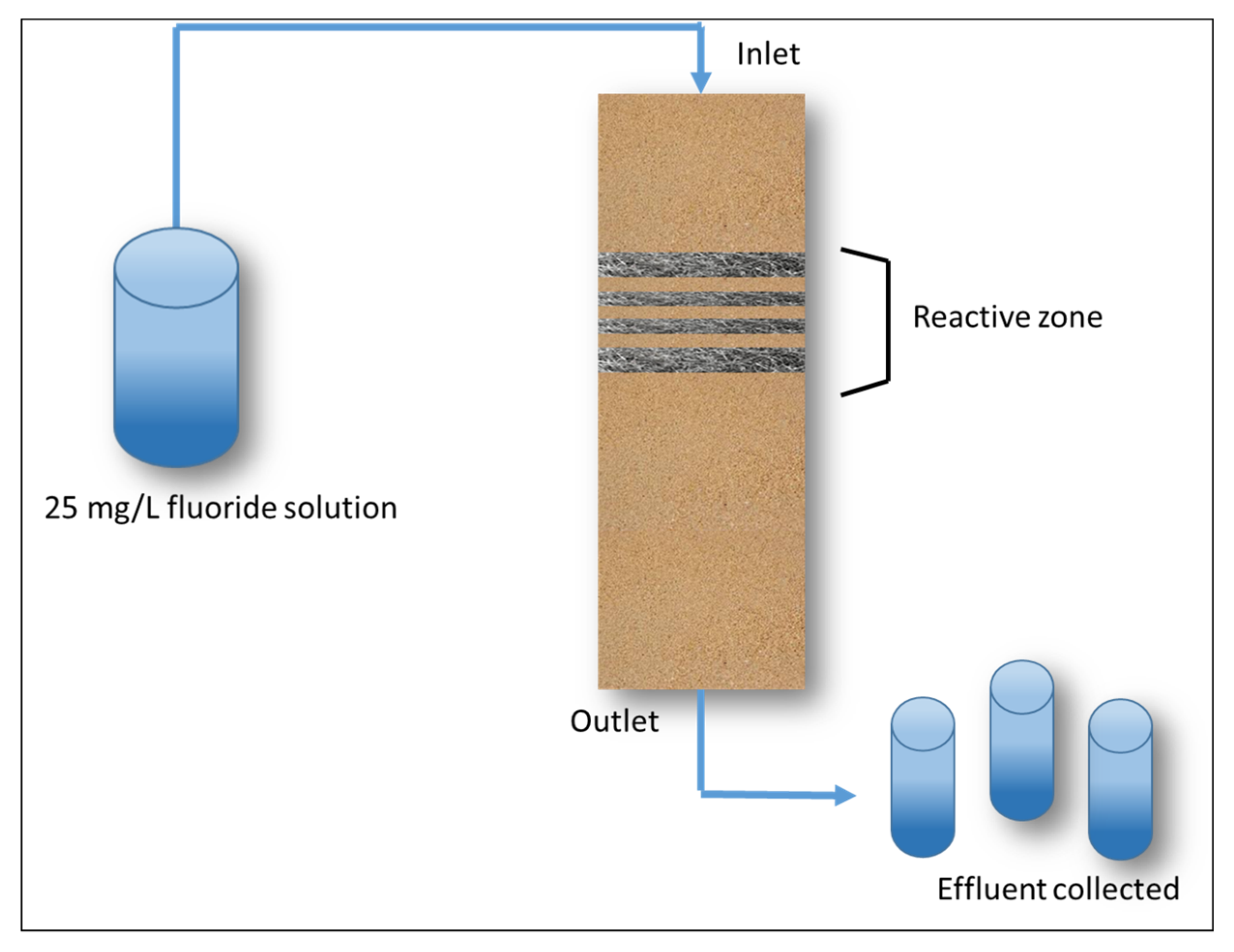
Processes Free Full Text Steel Wool For Water Treatment Intrinsic Reactivity And Defluoridation Efficiency Html
Remove the bulk of the dirt and rust with a stiff wire brush or steel wool.

. Some steel wool and a 9-volt battery will do the trick. When copper is displacedwe can then remove the separate the iron sulfate solution from the solid copper. After reaction chemical equation Fe CuSo4 FeSo4 Cu steel wool copper sulfate Iron ii sulfate copper Florence.
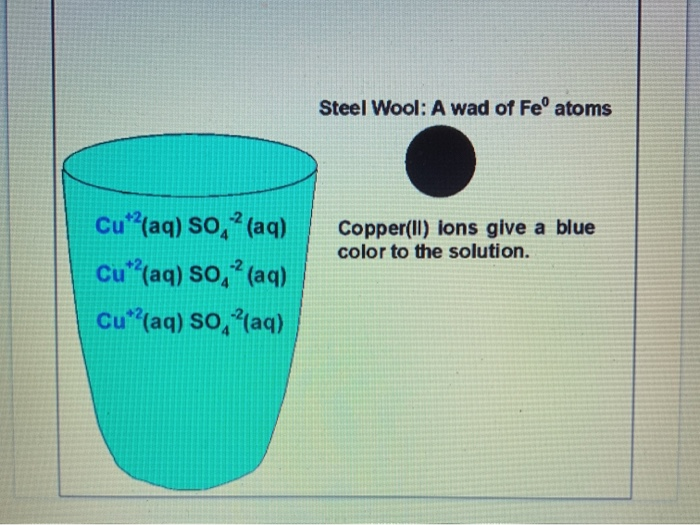
Before disposal these compounds must be treated to reduce their toxicity. Describe how to use steel wool which is made mostly of iron to remove the Cu 2 ions from an aqueous solution of copper II sulphate. Describe how to use steel wool which is made mostly of iron to remove the Cu2 ions from an aqueous solution of copperll sulfate.
If it is too large to fit in a beaker use a glass tub or tray. After Reaction A chemical property is the ability of a substance to change or react and. The fuzzy silver coating in Figure 3 of this section is impure silver.
This solution will need at least 48 hours to form and is needed for the demonstration. Describe how to use steel wool which is made mostly of iron to remove the Cu 2 ions from an aqueous solution of copperII sulfate. B Copper compounds such as copperll sulfate are toxic.
Make iron II acetate 1 -2 days prior to using the demonstration. Put the material in a glass beaker. Describe how to use steel wool which is made mostly of iron to remove the Cu2 ions from an aqueous solution of copperll sulfate.
Rust is the result of a chemical reaction between iron and oxygen otherwise known as iron oxide. Steel wool is available in several different strand thicknesses start with the largest thickness and work your way down to the fine one. Due to the fact that the iron in the steel wool chemically reacts with the aqueous solution we can conclude that during the reaction the iron displaces the copper.
Be sure not to remove any copper from the beaker. Before disposal these compounds must be treated to reduce their toxicity. Place the glass container outside or underneath a fume hood before proceeding.
Actually it is called a Galvanic Reaction or Galvanic Corrosion. Before disposal these compounds must be treated to reduce their toxicity. B Copper compounds such as copperll sulfate are toxic.
Add some kindling and you have the start of a fire. Before disposal these compounds must be treated to reduce their toxicity. Nails and paper clips are made of steel an alloy primarily composed of iron.
Carefully break up any clumps of copper with a spatula if drying proves difficult. B Copper compounds such as copperll sulfate are toxic. C are the best.
Steel touching copper water pipes can lead to electrolysis. When you add heat as from a. The saltvinegar solution dissolves some of the iron and its oxides on the surface of the nail leaving a negative charge on the surface of the nail.
Just touch the positive and negative terminals of the battery to the steel wool and it will start to glow and smolder the 9-volt battery is sending a current through the thing strands of steel wool. Describe how to use steel wool which is made mostly of iron to remove the Cu2 ions from an aqueous solution of copperll sulfate. Since steel wool-- and steel in general -- is largely made of iron steel wool is prone to rust if it doesnt have a rustproof coating on itThe actual reaction that creates rust happens when two iron atoms mix with three oxygen atoms in water.
Describe how to use steel wool which is made mostly of iron to remove the Cu2 ions from an aqueous solution of copperll sulfate. B Copper compounds such as copperll sulfate are toxic. To remove the tarnish from silverware soak it in a hot solution of baking soda in an aluminum pan.
Steps 2 and 3 describe how to make iron II acetate from white vinegar and steel wool. Describe how to use steel wool which is made mostly of iron to remove the Cu2 ions from an aqueous solution of copperll sulfate. Use 0000 steel wool shown here so it breaks down even faster in the vinegar.
Before disposal these compounds must be treated to reduce their toxicity. Put the object in a bucket of kitty litter then put the bucket on an oscillating object like an orbital sander to shake up the kitty litter and remove debris from the object by abrading. However it exists in the saltvinegar solution as positively charged copper ions as opposed to neutral copper metal.
In this case the copper would be. Add your copper coated material to a glass beaker. Use scissors to cut the 0000 grade steel wool from the bundle and then measure the mass to get 20 grams.
For each run calculate the moles of iron used and the moles of copper formed. Plastics and many metals will. Soak some steel wool in white vinegar for a few hours or a few days the longer it steeps the darker the aged effect will be.
No acid is usedIn the subtractive process for making PC boards one of two etchant salt solutions will be used to remove copper from where traces are not desiredferric chlorideammonium. B Copper compounds such as copperll sulfate are toxic. Fill the jar with the steel wool and vinegar the measurements dont have to be precise.
Fe s CuSO 4 aq - Cu s FeSO 4 aq the copper would form a precipitate and could be filtered from the solution FeSO 4 aq is non-toxic and actually used as a nutritional supplement. The oxygen bonds to the metal and a new. Over time this can destroy the integrity of copper piping.
Procedure 5 Https Youtu Be Kmhd8bmeflo Fill A Chegg Com

Homax 3 25 In X 4 In Coarse Steel Wool In The Steel Wool Department At Lowes Com

Simple Chemical Reaction Experiment Steel Wool And Vinegar Reaction
Comments
Post a Comment